T-MSC
T-MSCs are derived from ImStem’s patented technology, which can differentiate human embryonic stem cells (hESCs) into mesenchymal stem cells (MSCs) with high efficiency, purity, and consistency, through a trophoblast-like intermediate stage. The MSCs produced using this proprietary technology is named T-MSCs.
Our proprietary T-MSC technology enables us to achieve large-scale MSC production as well as consistency in MSC quality. T-MSC therapies contain MSCs differentiated from well-established human embryonic stem cell lines, therefore, avoiding donor-related variances and ethical issues that have been challenges for tissue-derived stem cell products. Another key feature of T-MSC is its low immunogenicity, or ability to evade immune surveillance, which means that T-MSC therapies can be administered with reduced/or no immune suppression treatment. Patients receiving T-MSC therapies would face a lower risk of immune rejection. All these advantages make T-MSC a first-in-class cell therapy to fulfill clinical requirements for cell-based therapies.
-
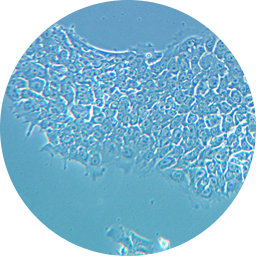
hESCs
-
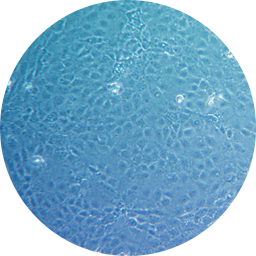
Trophoblast-like intermediate stage
-
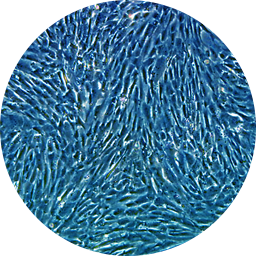
T-MSCs
ImStem’s Chief Technology Officer, Dr. Xiaofang Wang, and Dr. Renhe Xu are the inventors of T-MSC technology.

To date, T-MSC technology has been granted patents from important markets including the United States, Japan, Australia, China, and the European Union.
The first T-MSC technology-based cell therapy product, IMS001, was granted United States Food and Drug Administration (FDA) Investigational New Drug (IND) clearance, in March 2020, to treat multiple sclerosis. Since then, ImStem has been making progress in clinical studies. In the third quarter of 2021, the first patient was screened for the phase 1/2a Clinical Trial of T-MSC in multiple sclerosis in the United States. In the clinical studies, IMS001 is designed to administer intravenously without the need of donor matching.
Groundbreaking Science
T-MSC demonstrates three essential characteristics which make it an ideal “off-the-shelf” cellular medicine: consistency, safety, and effectiveness.
T-MSCs originate from allogeneic pluripotent human embryonic stem cells, which can be expanded in vitro almost indefinitely, and enables a scaled-up production of cell products for commercial purposes. ImStem owns its proprietary T-MSC technology, which ensures manufacturing homogenous cells with shorter differentiation time, greater efficiency, and increased consistency.
The preclinical safety profile of T-MSC is established through extensive preclinical GLP toxicology, biodistribution, and pharmacology studies.
T-MSC has a clear advantage over conventional small molecule medicine, as it may not just address symptoms of targeted diseases, but has the potential to repair damaged tissues and consequently improve or completely restore functions.
T-MSC Demonstrates Compelling Characteristics for Therapeutic Potential
This conclusion is based on the preclinical studies completed by ImStem compared to MSCs derived from adult tissues.
Clinical Applications of T-MSC
T-MSC based stem cell therapy has the potential to alleviate symptoms, restore tissue damage, improve functional outcomes, as well as potentially induce remission. On principle, we will design distinct cell products which are optimized in targeting each disease based on its specific pathologies. ImStem's clinical development programs are focused on treating a wide variety of degenerative, autoimmune, and inflammatory diseases where the current treatments are limited for many patients.
To learn more about the clinical trial of our T-MSC based cell therapy,
